LAWS OF CHEMICAL COMBINATION
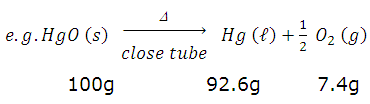
I.Law of constant composition or proportion : A definite chemical compounds always contain elements chemically combined in the same proportion by weight.
II.Law of multiple proportions : When two elements combine in more than one proportion i.e . to form more than one compound there is always the simple relationship between the fixed weight of one element and the weight of the other element present .
III.Laws of combining weight: Elements combined in the ratio of their combining weights or chemical equivalents; or in some simple multiple or sub-multiple of that ratio.
Also called the LAW OF RECIPROCAL PROPORTIONS or LAW OF EQUIVALENTS.
Distribution law of multiplication: The multiplication of a compound expression by a factor is the sum of the partial products of each term of the expression by that factors.Thus
(a +b + c) d =ad + bd + cd.
Hook`s Law : within the limits of elasticity , the stress is proportional to strain .
Law of Reciprocal proportion :See laws of chemical composition .
Law of reflection of light : (i) The angle of reflection is equal to the angle of incidence.
(ii) The incident ray, the reflected ray, and the normal to the reflecting surface at the point of incidence lie in the same plane.
Law of equivalence of mass and energy: See Einstein’s Law of the Equivalence of Mass and Energy.
Photo-electric effect: It is the phenomenon of emission of electrons by the substance when irradiated with light of a frequency greater than a certain minimum threshold frequency.
LAWS OF THERMO-DYNAMICS
First law of thermo-dynamics: The heat produced in a mechanical work is in proportion to the work done.
Lenz’s law: When a circuit and a magnetic field move with respect to each other, the direction of the induced current is such so as to oppose the motion to which it is due.
Inverse square law of illumination: The intensity of illumination of a surface by a point source of light varies directly with the illuminating power of the source and inversely as the square of the perpendicular distance from the source of the surface.
Inverse square law of electrical charges: See Coulomb’s Law.
The force between two poles varies inversely as the square of the distance between them.

