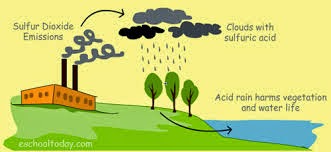
Acid rain is the common name for acidic deposits that fall to Earth from the atmosphere. Acid rain is rainfall that has been acidified. Rain is acidified by oxides of sulfur and nitrogen. Facts About Acid Rain you never knew is that Acid rain is formed when pollutants called oxides of sulfur and nitrogen, contained in power plant smoke, factory smoke, and car exhaust, react with the moisture in the atmosphere.
Read Also: Most Polluted Cities in India
Shocking Facts About Acid Rain You Never Knew
- The term was coined in 1872 by the Scottish chemist Robert Angus Smith (1817–1884) to describe the acidic precipitation in Manchester, England.
- Acid rain can also be produced from volcanic eruptions, burning coal and even rotting plant life.
- Acid rain cannot rot your skin. It usually doesn’t taste or smell any different than normal rain.
- The sulfur dioxide and nitrogen oxide that creates acid rain can cause diseases such as cancer, asthma, and even heart disease. It’s a concern in the air, but not in the rain itself.
- The acid in acid rain can damage a car’s paint job, but it won’t melt the car.
- Acid rain can actually kill a forest. The acid rain can kill the leaves on the trees by cutting off their light and nutrient supply. It also changes the acidity in the soil, making it impossible for trees and other plant life to grow. It also poisons the soil and plant life.
- When acid rain lands in the water such as streams, lakes and rivers, it changes the pH and makes the water toxic to the fish and other life in the water.
- Facts About Acid Rain you never knew is that entire lakes have been declared dead because of acid rain.
- Acid rain has a pH of 4.3 while pure water is perfectly balanced at 7.
- Acid rain has the same approximate pH as vinegar and orange juice.
- Rain is not the only type of precipitation that can be called acid rain. Snow, fog, and even dust can contain the same damaging toxins as acid rain.
- Acid rain can be neutralized the same way as the acid can be. In some environments, acid rain is more problematic. For instance, Eastern Canada lacks a natural alkalinity. Lime is able to neutralize the acid, but there is no lime in the ground in some areas and because of this the acid rain is able to do more damage.
- Sulfur dioxide, which is a major contributor to acid rain, is produced by burning fossil fuels and it is a by-product of many industrial processes.
- Facts About Acid Rain you never knew is that a large amount of the acid rain that reaches Canada is the result of emissions in the United States.
- Nitrogen oxide, a major contributor to acid rain, is produced by the exhaust from vehicles, from furnaces and other equipment. A large amount of Canada’s nitrogen oxide emissions originates in the United States.
- Despite major efforts to decrease acid rain, it is still killing lakes and aquatic life. 95,000 lakes in North America have been damaged by acid rain.
- In high-acid environments, lake waters grow unnaturally clear, as plankton and other types of microscopic
life succumb. The reproduction of aquatic animals is hindered or stopped. Then, too, aluminum and other metals, normally found in harmless compounds, are released from the soil in toxic forms. The aluminum attacks the gills of fish, making breathing difficult. They literally suffocate.
- Facts About Acid Rain you never knew is that Coal is also the source of a worse culprit, sulfur dioxide, which makes up the other 70 percent of acid rain. The annual discharge of these gasses into North American skies is 60 million tons. The atmosphere is becoming a garbage dump.
- In North America, 30 percent of acid rain is caused by nitric oxides-half of which comes from motor vehicle
exhausts. The other half comes from the burning of fossil fuels, by electric utilities and other industries.
Must Read:
General Knowledge Facts File – Important


Supeb_/_.-.-.-.-.-.-.-.-.-.-.-.-